23 August 2017
Researchers investigate the lighter side of Earth’s inner core
Posted by Lauren Lipuma
By Patricia Waldron
Scientists can generally agree that Earth’s inner core, located more than 5,000 kilometers (3,000 miles) beneath the surface, is a dense ball of iron with a small amount of nickel. It likely contains lighter elements as well, such as silicon, oxygen, sulfur, carbon, or hydrogen, but researchers have yet to come to a consensus on the identities and quantities of these lighter elements.
Researchers report in a new study that a carbon compound called iron carbide, combined with small amounts of silicon impurities, may be an important component of the inner core. The researchers performed computer simulations to model how an iron and nickel core containing either iron carbide, or iron carbide with some silicon, compares to the density and other known characteristics of the inner core. Adding silicon to the simulation gave the best alignment with Earth’s actual characteristics. The researchers report their findings in a new study in Geophysical Research Letters, a journal of the American Geophysical Union.
“We are trying to simulate the conditions deep inside Earth, to understand the evolution and composition of the core,” said Sujoy Ghosh, a researcher at the Indian Institute of Technology in Kharagpur, India and co-author of the new study. “Because carbon is abundant and can dissolve in iron, we suggest that carbon is a probable light element in the core. The mantle surrounding the core is rich in silicon and, under the local temperature and pressure conditions, some of that silicon could become incorporated into the core. Silicon and carbon atoms have the same charge, so silicon could easily substitute for carbon in iron carbide.”
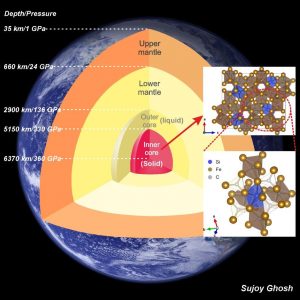
Internal structure of Earth showing the liquid outer core and solid inner core. Silicon-doped iron carbide, shown at the right-hand side of the diagram, can be a possible carbide phase for the inner core, coexisting with iron. The silicon atoms are blue, the carbon is brown, and the iron is represented by gold circles.
Credit: Sujoy Ghosh.
Researchers initially put forth a form of iron carbide called cementite, which has three molecules of iron for every one molecule of carbon, as a potential component of the inner core in the mid-1990s. Computer simulations later ruled out the idea because that form is too stiff at high pressures. More recently, researchers have suggested that iron carbide with seven iron atoms for every three carbon atoms is a better candidate. Iron carbide with this composition has two possible arrangements, a square-shaped orthorhombic phase and a hexagonal phase, and scientists disagree about which form occurs in the inner core.
Ghosh partnered with Swastika Chatterjee, a physicist colleague at the Indian Institute of Technology Kharagpur, to perform computer simulations of how the two phases of iron carbide, with and without silicon impurities, would behave inside Earth’s inner core under appropriately high pressure and temperature conditions. Previously, no one had explored the possibility of silicon and carbon joining within the same compound to make up the lighter portion of the core..
The researchers compared their results to the Preliminary Reference Earth Model, which represents average Earth properties, including density and the way sound waves generated by earthquakes, called seismic waves, propagate through the core. Much of what we know about Earth’s core comes from studies measuring how seismic waves pass through the layers of the planet. The researchers observed that when they “doped” the iron carbide by replacing a small percentage of the carbon atoms with silicon, the resulting core material had greater agreement with Earth’s properties at inner core conditions. They also noticed that the silicon-doped orthorhombic phase produced better agreement than the hexagonal phase of iron carbide.
If the researchers are correct, then Earth’s core is a sizable reservoir of carbon and silicon within the planet, as proposed previously by several researchers. Ghosh is already exploring these results further in the lab, in collaboration with colleagues at Tohoku University and Indian Institute of Science Education and Research-Kolkata. He has combined silicon, iron, and carbon inside an apparatus called a DIA-type multi-anvil apparatus, which can generate extreme pressures. Very soon, however, he hopes to perform this type of experiment at his home institution. Ghosh has already ordered one high-pressure instrument, an end-loaded piston cylinder apparatus, and hopes to acquire a second, a multi-anvil apparatus, soon to complete his lab set up.
“In the Earth sciences community in India, we don’t have high-pressure facilities,” Ghosh said. “This is a huge task, but if I can build a high-pressure lab here, then this would be a big contribution for my country.”
— Patricia Waldron is a freelance writer. This post originally appeared as a feature story on the Deep Carbon Observatory website.










 GeoSpace is a blog on Earth and space science, managed by AGU’s Public Information staff. The blog features posts by AGU writers and guest contributors on all sorts of relevant science topics, but with a focus on new research and geo and space sciences-related stories that are currently in the news.
GeoSpace is a blog on Earth and space science, managed by AGU’s Public Information staff. The blog features posts by AGU writers and guest contributors on all sorts of relevant science topics, but with a focus on new research and geo and space sciences-related stories that are currently in the news.
I think this is excellent work by research geophysicists of IIT KGP. This is the sort of work that enhances the reputation of an academic institution. Personally, I am quite thrilled that the Department of Geology and Geophysics, IIT KGP, my alma mater is procuring extreme HP equipment soon. Dr. A.B.Mukherjee used to study phase behaviour in a high pressure lab earlier, but this equipment will far supercede the pressures that could be attained at that te.